- Share this article
- Subscribe to our newsletter
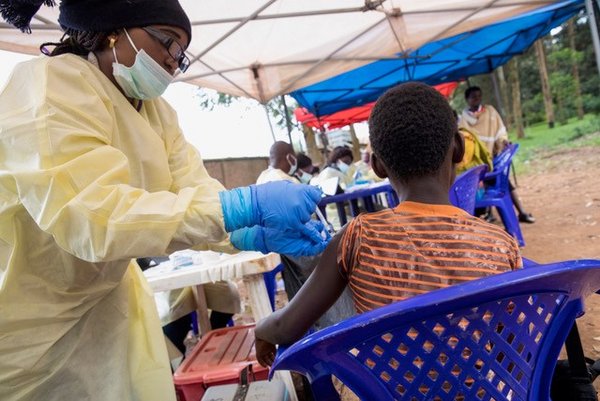
A vaccine to prevent Ebola
For the first time, an Ebola vaccine has been prequalified by the World Health Organization (WHO) in November 2019. This is a critical step that will help speed up its licensing, access and roll-out in countries most at risk of Ebola outbreaks. It is the fastest vaccine prequalification process ever conducted by WHO.
“Prequalification” means that the vaccine meets WHO standards for quality, safety and efficacy. United Nations agencies and Gavi, the Vaccine Alliance, can procure the vaccine for at-risk countries based on this WHO recommendation.
Two days earlier, the European Commission decided to grant a conditional marketing authorisation for the vaccine, following the recommendation from the European Medicines Agency (EMA). Representatives from the prequalification team participated in the EMA evaluation process to address programmatic suitability for at-risk countries in Africa.
The injectable Ebola vaccine, Ervebo, is manufactured by Merck (known as MSD outside the US and Canada). It has been shown to be effective in protecting people from the Ebola Zaire virus and is recommended by the WHO Strategic Advisory Group of Experts (SAGE) for vaccines as part of a broader set of Ebola response tools. The decision is a step towards greater availability of the vaccine in the future, though licensed doses will only be available as of mid-2020.
WHO is also facilitating licensing of the vaccine for use in countries at risk of Ebola outbreaks, based on the reviews and positive outcome by EMA. WHO, with the support of EMA, has worked closely with many African regulators who have indicated they will quickly license the vaccine following the WHO recommendation.
(WHO/ile)
Read more at the WHO website: https://www.who.int/news-room/detail/12-11-2019-who-prequalifies-ebola-vaccine-paving-the-way-for-its-use-in-high-risk-countries




Add a comment
Be the First to Comment